Why Do I Wake Up at 3 AM Every Night? Causes by Mechanism
Cortisol typically rises a few hours before habitual wake time. REM sleep dominates the second half of the night. So 3 AM waking isn't unusual — but what sleep research describes about why you can't get back to sleep is what to investigate.
This is an informational summary of peer-reviewed sleep research and clinical literature. It is not medical advice, diagnosis, or treatment recommendation. The mechanisms and self-relevant markers described below are intended to inform a conversation with a clinician — not to substitute for medical evaluation. If sleep difficulties affect your daily life, please consult a licensed healthcare professional. For self-harm or suicidal thoughts, contact emergency services or a crisis line immediately.
3:14 a.m. Not the bathroom. Not the alarm. You just woke up. You check the clock. You close your eyes. Yesterday's meeting starts looping. You drift back around 4. The 6:30 alarm goes off. You wake up more tired than yesterday.
And then you search — "why do I wake up at 3 a.m. every night?" The first page of results splits in two. Half is about spiritual meaning or the traditional Chinese-medicine "organ clock" (liver → lungs → large intestine). The other half is generic "stress and caffeine." This article is in the second category — but rather than a generic listicle, it organizes the causes by mechanism, drawing on clinical sleep medicine literature.
The key facts. Two physiological systems put 3 a.m. on the wake-promoting slope of the night. (1) The cortisol awakening response — Bowles and colleagues' 2022 study in Frontiers in Neuroscience reported that cortisol peaks at the circadian phase corresponding to about 3:40 a.m. in their controlled protocol — that is, on the body's internal clock, not necessarily clock time, and typically several hours before habitual wake. (2) REM sleep dominates the second half of the night, leaving the second half lighter and easier to wake from than the first half, as described in standard sleep physiology references. So 3 a.m. waking is on the body's wake-promoting slope, not a malfunction. The clinically meaningful question is: why can't I get back to sleep, and is it happening every night?
This article walks through (1) what is actually happening in the body around 3 a.m., (2) circadian causes, (3) pharmacological causes, (4) physiological causes, (5) psychological causes — each with markers worth bringing to a clinician, (6) when research describes escalation, and (7) lifestyle adjustments that show up in sleep guidance literature.
This article is an informational summary of clinical sleep medicine literature, not individual medical advice. Please consult a clinician before applying any of this to your own situation. The article does not engage spiritual or traditional-medicine framings of 3 a.m. waking — those are a different conversation.
The 3 AM window — what your body is doing
Sleep cycle structure
Sleep cycles repeat in roughly 90–120 minute units, with each cycle progressing through NREM stages (N1 → N2 → N3) and into REM. As described in standard physiology references, the first half of the night is dominated by N3 (deep slow-wave sleep), while the second half is dominated by REM. REM episodes lengthen toward morning. The structure is described in more detail in How Sleep Cycles Work.
REM sleep produces a brain state that is electrophysiologically close to wakefulness — active brain, paralyzed body. From this state, small disturbances (a noise, a temperature shift, a bladder signal) are described in the literature as more likely to convert into a full awakening than they would be from N3.
Cortisol awakening response (CAR)
Cortisol does not stay flat across the day. It follows a circadian profile, peaking near the end of the night and falling to its lowest point in the evening. Bowles 2022 reported that, under a controlled circadian protocol, the CAR peaked at the circadian phase corresponding to roughly 3:40 a.m. — a phase several hours before habitual wake time. The cortisol rise is described in the literature not as the cause of waking but as part of a physiological cascade preparing for morning activity.
Putting these together: 3 a.m. is a window where (a) REM dominates and the brain is easier to wake from, and (b) cortisol is on its rising slope. A small disturbance plus a light brain state plus a rising arousal hormone can produce an awakening that feels surprising. The clinical literature consistently describes this kind of late-night waking as common rather than unusual; what tends to be clinically interesting is the inability to return to sleep, especially when it happens nightly.
For prevalence context, Ohayon's 2008 phone survey in Journal of Psychiatric Research (n=8,937 U.S. adults) reported that nighttime awakenings occurring three or more nights per week were experienced by a substantial fraction of the adult population — that is, frequent night waking is statistically common rather than rare.
Circadian causes — when the clock is fine but arousal is amplified
The CAR itself is normal physiology. The clinical literature describes amplitude of the cortisol response and overall arousal as elevated in stress, PTSD, and some forms of depression.
Early morning awakening — a depression marker
The DSM-5 includes early morning awakening with inability to return to sleep among the features of melancholic depression. Nutt, Wilson, and Paterson's 2008 review in Dialogues in Clinical Neuroscience reported that approximately three-quarters of patients with depression report sleep symptoms, with characteristic patterns including:
- Shortened REM latency — REM appearing earlier in the night than typical
- Increased REM density
- Early morning awakening with difficulty returning to sleep
If 3–4 a.m. waking has been near-nightly for two weeks or more and coincides with low mood, anhedonia, or appetite or weight changes, the clinical literature describes this combination as a marker for which a primary-care or mental-health evaluation is the typical next step.
Markers worth bringing to a clinician
If two or more of the following are present, the literature describes a primary-care or mental-health evaluation as a generally appropriate step:
- Waking at nearly the same time (3:00–4:30) most nights
- Difficulty returning to sleep after waking
- Low mood or loss of interest persisting for two or more weeks
- Changes in appetite, weight, or concentration
- Any thoughts of self-harm or suicide — contact emergency services or a crisis line immediately, regardless of other markers.
These markers are framed as information to bring into a conversation with a clinician — they are not a self-diagnostic tool.
Pharmacological causes — alcohol, hypnotics, caffeine, medications
Reversible causes — substance and medication-related — are among the most commonly described in the clinical literature.
Alcohol — the first-glass trap
The common belief that "a glass of wine helps me sleep" is partly accurate: alcohol does shorten sleep onset. The second half of the night, however, is a different story. Ebrahim and colleagues' 2013 review in Alcohol: Clinical & Experimental Research reported that moderate to high alcohol consumption is associated with reduced total nighttime REM and delayed first REM onset, along with increased sleep disruption and awakenings during the second half of the night. This pattern of late-night fragmentation has been consistently described as a documented effect of evening alcohol.
So the same drink that shortens sleep onset also tends to cause the 3 a.m. waking that follows. A common self-observation suggested in the literature is a one-week alcohol-free trial to see whether the waking pattern changes.
Hypnotic withdrawal
Long-term use of benzodiazepines (lorazepam, alprazolam) or Z-drugs (zolpidem, zaleplon) followed by abrupt discontinuation or rapid taper is associated with rebound insomnia in the clinical literature. Any change to a prescribed medication should be discussed with the prescribing clinician — discontinuation without medical guidance is not advised.
Caffeine — past noon
Caffeine has an average half-life of about 5 hours, with individual variation between roughly 1.5 and 9.5 hours (Institute of Medicine pharmacology reference, NCBI Bookshelf). Gardiner 2024 reported that 400 mg of caffeine four hours before bed extended sleep onset by an average of 14.2 minutes — and effects on overall sleep quality and night waking have been described in associated literature. An afternoon caffeine cutoff is the most common practical suggestion.
Medications often overlooked
Several medication classes are described in clinical literature as potentially associated with late-night waking:
- Beta-blockers (for example propranolol). Stoschitzky and colleagues' 1999 study reported that (S)-propranolol reduced nocturnal melatonin metabolite excretion by approximately 80%.
- Corticosteroids (for example prednisone) — cortisol-like effects are described as potentially increasing arousal.
- Diuretics taken late in the day — increased nocturnal urination (nocturia).
- Some SSRIs, particularly during initiation — sleep architecture changes are documented.
- Some stimulant medications (for ADHD, for example) — residual effect into the evening.
If a new awakening pattern began within 6–12 weeks of starting a new medication, a review with the prescribing clinician is the typical next step described in the literature. Do not change a prescribed medication on your own. The medication discussion in Why Am I Tired After 8 Hours of Sleep? covers similar ground from the daytime fatigue angle.
This is informational background only. Discussion of any prescribed medication should occur with the prescribing clinician.
Physiological causes — OSA, GERD, RLS, nocturia, menopause, pain
Cases where the body itself drives awakenings. Generally these warrant clinical evaluation.
Obstructive sleep apnea (OSA)
- Pattern: Awakenings are often brief microarousals that the person doesn't remember. The first signal often comes from a partner reporting loud snoring, witnessed breathing pauses, or gasping.
- Screening: The STOP-BANG questionnaire is a screening tool used in many clinical workflows. Pooled meta-analytic estimates (for example Nagappa 2015 in PLOS ONE) describe sensitivity in the 85–90% range for any OSA at AHI≥5 — meaning it is good at flagging cases for further evaluation.
- Diagnosis: STOP-BANG is a screener, not a diagnostic test. A sleep study (in-lab polysomnography or a home sleep apnea test) is what produces a diagnosis, as described in clinical guidelines.
Nocturia (nighttime urination)
Waking once a night to urinate is common; clinical urology literature often describes two or more episodes per night as worth evaluating. Common contributors include OSA (frequently underrecognized), prostate issues, diabetes, evening fluid intake, and diuretic timing. Reducing fluid intake after 6 p.m. and shifting any diuretic dose earlier (in consultation with the prescriber) are frequently described initial steps.
Restless legs syndrome (RLS)
Unpleasant leg sensations in the evening that are temporarily relieved by movement. Sleep disruption from limb movement during the night is a documented feature. The 2021 Mayo Clinic Proceedings algorithm from the RLS Foundation Medical Advisory Board describes ferritin below approximately 75 ng/mL with transferrin saturation below 45% as a threshold at which an oral iron trial is generally considered. Evaluation by a sleep medicine or neurology clinician is the standard pathway described.
GERD (gastroesophageal reflux)
Reflux when lying down can cause early-morning waking with chest burning, sour taste, or coughing. Avoiding food in the three hours before bed, elevating the head of the bed, and avoiding common triggers (spicy, fatty, caffeinated, alcoholic) are typically recommended initial steps in the literature.
Menopause and perimenopause
Hormonal changes and hot flashes are described as commonly disrupting late-night sleep, often in the early morning hours. Hormone therapy and other options are discussed in the gynecology and sleep literature; the specific approach is a clinical conversation.
Chronic pain
Reductions in N3 (deep) sleep and increased nocturnal awakenings have been documented across chronic pain conditions. Both the underlying pain and pain medications can affect sleep architecture.
Psychological causes — hyperarousal, anxiety, rumination
The hyperarousal model
Riemann and colleagues' 2010 review in Sleep Medicine Reviews proposed hyperarousal as a core mechanism of chronic insomnia. The model describes a combination of genetic vulnerability and external stressors leading to elevated activation across cortical, autonomic, and neuroendocrine systems — an arousal that doesn't shut down at bedtime and reactivates at night-time awakenings.
Symptoms commonly described:
- Faster heart rate, warmth, or sweating after waking at night
- Racing thoughts that resist redirection — yesterday's meeting, tomorrow's deadline, an old mistake
- The sensation of being physically tired but cognitively activated
Conditioned arousal
Waking, checking the clock ("3 a.m. again — how am I going to sleep now?"), and the resulting amplification of arousal can become a learned pattern over repeated nights. Over time, the bed itself can become associated with arousal rather than sleep, as described in the conditioning literature.
Rumination
Darkness removes external distractions. Internal stimuli — thoughts — fill the gap. The same mild concern that wouldn't register at noon can feel catastrophic at 3 a.m. — a documented cognitive pattern often referred to as nocturnal rumination.
Markers worth bringing to a clinician
- Elevated heart rate or sweating immediately after waking
- Inability to redirect racing thoughts
- An immediate "I won't get back to sleep" reaction on seeing the clock
- Daytime anxiety or a persistent sense of being "always on"
When this pattern is consistent, CBT-i — particularly stimulus control and cognitive components — is described in clinical guidelines as the typical first-line approach for chronic insomnia. Falling asleep specifically is covered in the sister post How to Fall Asleep Faster: What Research Shows.
These markers are framed as information for a clinician, not a self-diagnostic checklist.
When research describes escalation to a clinician
DSM-5 chronic insomnia disorder
The criteria describe chronic insomnia as:
- Difficulty initiating sleep, maintaining sleep, or early morning awakening
- Occurring three or more nights per week, for three or more months
- Despite adequate sleep opportunity
- With daytime impairment (fatigue, attention, mood, work, or school effects)
When all four are present, the clinical literature describes evaluation by a primary-care clinician or sleep specialist as the typical next step.
Reasons not to wait three months
- A bed partner reports loud snoring, witnessed breathing pauses, or gasping → screening followed by sleep study (OSA)
- A bed partner reports significant limb movements → evaluation for RLS or PLMS
- Early morning awakening combined with low mood for two or more weeks → depression evaluation; tools such as PHQ-9 are used in some clinical settings
- Any thoughts of self-harm or suicide → contact emergency services or a crisis line immediately
- Daytime sleepiness producing microsleep events while driving → urgent evaluation
- Pain, GERD, prostate symptoms or other physical drivers → evaluation of the underlying condition
- A new awakening pattern beginning within 6–12 weeks of starting a new medication → review with the prescribing clinician
What clinicians typically describe
- CBT-i is generally described as first-line for chronic insomnia in the AASM 2021 guideline and other major guidelines.
- Pharmacologic treatment is addressed in a separate AASM guideline (Sateia et al., 2017) and is typically described as a secondary option rather than a substitute for CBT-i.
- Suspected physical causes (OSA, RLS, and others) are typically evaluated and treated separately — behavioral approaches are not described as a substitute for treatment of the underlying condition.
The escalation patterns summarized here describe what clinical guidelines lay out. Whether a specific situation meets these criteria, and what the appropriate evaluation looks like, is determined by a clinician.
Lifestyle adjustments described in sleep guidance
Reversible behaviors that show up frequently in sleep guidance literature:
- Avoid clock-watching. Conditioned arousal patterns are reinforced by checking the time. Turning the clock face away (or relying on the alarm only) is a commonly described practice.
- The 20-minute rule (stimulus control). If sleep doesn't return within roughly 20 minutes after waking, leaving the bedroom and returning only when sleepy is the standard stimulus-control practice. This is described as helping rebuild the "bed = sleep" association.
- A one-week alcohol-free trial. A frequently described self-observation step — does the waking pattern change?
- A noon caffeine cutoff. Given the average 5-hour half-life and individual variation up to 9.5 hours.
- Reduced fluid intake after 6 p.m. If nocturia is a contributor.
- Bedroom temperature near 18.3 °C (65 °F). Described as supporting sleep maintenance, not just sleep onset.
- Consistent wake time. Windred 2024 in SLEEP (n=60,977 UK Biobank) reported sleep regularity as a stronger predictor of all-cause mortality than sleep duration. Translated to night waking, a consistent schedule is described in the literature as associated with reduced fragmentation. Cycle-aligned bedtimes for various wake times are worked out in What Time Should I Go to Bed if I Wake Up at 6 AM?.
- Weekend lie-ins of one hour or less. Weekend recovery sleep is not described in the recent literature as a fix for chronic short sleep — see Can You Catch Up on Sleep on Weekends? for the underlying evidence.
What ties these together: the goal described in the literature is not the elimination of nighttime awakenings — those are part of normal sleep architecture. The goal described is the ability to return to sleep quickly after waking, a capacity that research consistently associates with consistent schedule, environment, and behavioral patterns.
These lifestyle items summarize what frequently appears in sleep guidance literature. Specific medical situations — current medications, chronic conditions, shift work — change how they apply, and a clinical conversation is the typical next step.
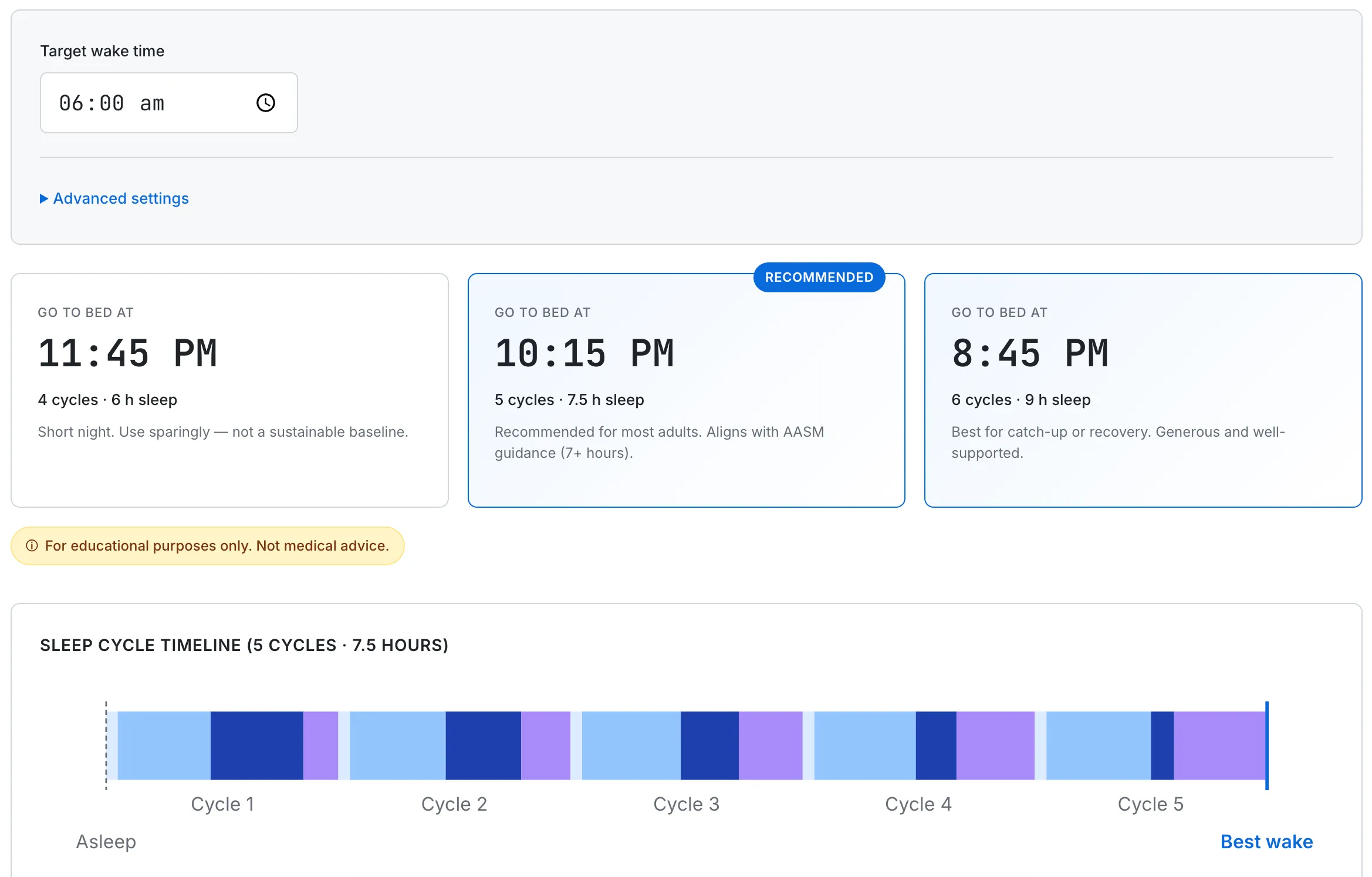
Research describes consistent schedule as associated with reduced nighttime fragmentation. The calculator helps you set a regular bedtime around a fixed wake time — informational support, not a treatment for an underlying cause.
This article summarizes peer-reviewed research and clinical literature for informational purposes only. It is not medical advice, diagnosis, or treatment recommendation. Conditions including obstructive sleep apnea, depression, and restless legs syndrome require evaluation by a licensed clinician — the markers described here are intended to inform a conversation with a healthcare professional, not to substitute for medical assessment. For self-harm or suicidal thoughts, contact emergency services or a crisis line immediately. If you have ongoing sleep difficulties, please consult a licensed healthcare professional. Information here represents the published literature as of April 2026 and may be revised as guidelines and evidence evolve.