How to Reset Your Sleep Schedule: A Practical Guide
The single all-nighter "reset" is a myth. The real reset is light and low-dose melatonin timed by the phase response curve. The mechanism-first guide.
This is an informational summary of circadian science and the published evidence on how to reset your sleep schedule, drawn from PubMed/PMC, AASM, NIH, CDC, and peer-reviewed chronobiology research. It is not medical advice, diagnosis, or treatment guidance. The studies cited describe controlled trials and population data — not personal predictions, and individual circadian response varies with age, chronotype, medications, light exposure history, and underlying conditions. For persistent schedule misalignment, suspected delayed/advanced sleep phase disorder, shift work sleep disorder, or non-24 sleep-wake disorder, please consult a licensed sleep-medicine professional.
The most common myth about how to reset your sleep schedule is the single all-nighter — stay awake for 24–36 hours, then sleep at the new desired time, and the body clock should "snap" into place. It does not work. Twenty-four hours of wakefulness raises homeostatic sleep pressure (adenosine accumulation) so that the next night's sleep comes faster — but the molecular clock in the suprachiasmatic nucleus (SCN) is essentially unmoved by sleep deprivation alone. Without aligned light and melatonin signals, the body drifts back toward its prior schedule within days. The actual reset, well-documented across half a century of chronobiology, is a phase shift driven by zeitgebers — primarily light, secondarily low-dose melatonin — applied at the right circadian time. Khalsa and colleagues' 2003 light phase response curve study showed that a single 6.7-hour 10,000-lux light pulse, delivered at the right time, can move the human clock by up to −3.60 hours (delay) or +2.01 hours (advance). The same light at the wrong time moves the clock the wrong way. So how to reset your sleep schedule is, fundamentally, a question of timing, not willpower.
The short answer. Resetting your sleep schedule means aligning light exposure (the strongest zeitgeber) and low-dose melatonin (the secondary zeitgeber) with the direction you need to shift, per the phase response curve. The human SCN's intrinsic period averages 24.18 hours (Czeisler et al., 1999, Science), so the natural drift without zeitgebers is later. To wake earlier (phase advance) — direct sunlight or 10,000-lux light immediately on waking, avoid evening light, optionally low-dose melatonin (≈0.3–0.5 mg) in the late afternoon. To stay up later (phase delay) — bright light in the evening, dim mornings. Adaptation rates from CDC's authoritative travel-medicine guidance run roughly 1 hour per day eastward, 1.5 hours per day westward. The single all-nighter, untimed light boxes, high-dose melatonin (5–10 mg), and blue-blocking glasses do not, on the published evidence, reliably reset the clock.
This post walks through eight things — (1) what is actually being "reset" (the SCN mechanism), (2) why light is the dominant zeitgeber and how the phase response curve works, (3) why timing of melatonin matters more than dose, (4) common reset scenarios (jet lag, DST, social jet lag, schedule drift), (5) evidence-based intervention strategies, (6) four 7-day plan templates, (7) what does not work or is over-marketed, and (8) when clinical evaluation is described as warranted.
What is actually being "reset" — the SCN circadian system
The phrase "reset your sleep schedule" points to a real biological system — the suprachiasmatic nucleus (SCN), a paired structure of roughly 20,000 neurons in the hypothalamus that functions as the master circadian clock. The SCN receives light signals through intrinsically photosensitive retinal ganglion cells (ipRGCs) — a separate retinal pathway from the rods and cones used for vision. Without external time cues, the SCN's intrinsic rhythm averages 24.18 hours across both younger and older adults (Czeisler et al., 1999, Science). That ~11-minute deviation from a clean 24-hour day is small but cumulative: with no zeitgeber input, a person drifts to a later schedule each day.
Two concepts that recur throughout the rest of this post:
- Phase advance — moving the body clock earlier. Falling asleep and waking earlier. The relevant scenarios: needing to wake up earlier for school or work; eastbound flight; recovering from a "stayed up too late" pattern.
- Phase delay — moving the body clock later. Falling asleep and waking later. Less common as a personal goal, but the natural drift direction without zeitgebers; the relevant scenarios: westbound flight; weekend "sleep in" extending into Monday.
Melatonin operates as both an output of the SCN (released by the pineal gland in dim light, suppressed by bright light) and as an input back to the SCN (a hormone that can shift the clock when administered at the right time). The clinical phase marker most often used in sleep medicine research is Dim Light Melatonin Onset (DLMO) — the moment in the evening when melatonin secretion begins. Most interventions described below are timed relative to DLMO or to estimated clock time.
Light as the dominant zeitgeber and how the phase response curve works
Light is by far the strongest zeitgeber for the human SCN. The same light pulse can delay or advance the clock depending on when it is delivered relative to the body's circadian phase — captured graphically in what chronobiologists call the phase response curve (PRC):
- Light before the core body temperature minimum (typically 2–3 hours before habitual wake time, around 4 AM for many adults) produces a phase delay — the clock moves later.
- Light after the core temperature minimum produces a phase advance — the clock moves earlier.
Khalsa, Jewett, Cajochen, and Czeisler's 2003 paper in The Journal of Physiology mapped this curve experimentally with single 6.7-hour pulses of approximately 10,000 lux white light. Maximum measured shifts were phase delays of up to −3.60 hours and phase advances of up to +2.01 hours. A single carefully timed light exposure, in other words, can move the human clock by 1 to 3 hours. The same exposure at the wrong circadian phase produces an effect in the wrong direction or no useful effect at all. The 2015 American Academy of Sleep Medicine clinical practice guideline for circadian rhythm sleep–wake disorders describes typical clinical light therapy as 30 minutes to 2 hours, in the 2,500–10,000-lux range, timed to the desired shift direction.
An everyday consequence: indoor lighting is dramatically dimmer than people perceive. Typical indoor environments deliver only 100–500 lux at the eye, while overcast outdoor conditions are 1,000–10,000 lux and direct sunlight is 50,000–100,000 lux. Brown and colleagues' 2022 PLOS Biology consensus recommendation is a minimum melanopic equivalent daylight illuminance (EDI) of 250 lux at the eye during daytime and below 10 lux for the three hours before bed. Most adults, spending the majority of their hours indoors, get a much weaker day–night light contrast than the SCN evolved to expect. The first natural intervention for resetting a sleep schedule is therefore not a light box but stepping outside in the morning.
| Light timing | Effect on the clock | Magnitude (single 10,000-lux pulse) |
|---|---|---|
| Evening / early biological night (before core temperature minimum) | Phase delay | Up to −3.60 hours |
| Around core body temperature minimum (~4 AM for typical sleeper) | Transition zone (near zero) | ~0 |
| Late night to morning (after core temperature minimum) | Phase advance | Up to +2.01 hours |
| Afternoon | Low sensitivity | Minimal |
Why timing of melatonin matters more than dose
Melatonin works two ways — as a hypnotic (the sedating effect of higher doses) and as a chronobiotic (the phase-shifting effect of low doses delivered at the right time). The two effects have different optimal doses and different timings, and the popular conflation of them is the source of most misuse.
Burgess, Revell, and Eastman's 2010 study in The Journal of Clinical Endocrinology & Metabolism mapped the human melatonin phase response curve. Two important findings:
- 0.5 mg and 3.0 mg produced similarly sized phase shifts — maximum advance about 1.5 hours, maximum delay about 1.3 hours. Higher doses did not produce larger phase effects.
- Timing was decisive. Maximum advances occurred when 0.5 mg melatonin was taken in the afternoon, 2–4 hours before DLMO — roughly 9–11 hours before sleep midpoint. Doses taken earlier in the morning produced phase delays.
For practical purposes:
| Goal | Low-dose melatonin (~0.3–0.5 mg) | Light |
|---|---|---|
| Phase advance (sleep earlier) | Afternoon or early evening, ~5–7 hours before desired bedtime (≈2–4 hours before DLMO) | Bright light immediately on waking |
| Phase delay (sleep later) | Soon after waking | Bright light in the evening; darkness/dim morning |
High-dose melatonin (5–10 mg) is widely sold but does little additional phase work and tends to produce next-day grogginess, headache, and vivid dreams. Erland and Saxena 2017 measured the actual melatonin content of 31 commercial supplements and found that label values diverged from measured values by −83% to +478% — a roughly six-fold range above and roughly five-fold below the label. Some products also contained serotonin as a contaminant. Regulatory status varies internationally — over-the-counter in the United States, Canada, and Australia; prescription-only in the United Kingdom and several EU countries. This article describes the published evidence on timing and dose; it does not recommend specific products or doses, and a clinician's input is the standard professional path for individual situations.
Common reset scenarios — jet lag, DST, social jet lag, schedule drift
Jet lag. Direction matters more than distance. Eastward travel requires a phase advance — biologically harder, because the SCN's PRC produces smaller advance shifts than delay shifts at comparable light intensities, and because the natural drift without zeitgebers is toward delay. Westward travel requires phase delay — easier, because the body is going with its natural drift direction. CDC's Yellow Book 2026 chapter on jet lag disorder cites adaptation rates of approximately 1.5 hours per day for westward travel and 1 hour per day for eastward travel — meaning an 8-hour eastward shift takes roughly 8 days for full circadian re-entrainment, while the same distance westward takes about 5–6 days. The CDC chapter also describes 0.5–1 mg of melatonin as often sufficient for circadian shifting, and notes that 200 mg of caffeine every 4 hours (stopped at least 6 hours before desired bedtime) can be a useful adjunct for daytime alertness.
Daylight Saving Time transitions. The spring-forward shift is essentially a one-hour phase advance imposed simultaneously on entire populations, plus an acute hour of sleep loss. Manfredini and colleagues' 2019 review reports a "modest increase in occurrence of acute myocardial infarction, especially in the first week after the spring shift" associated with DST. The fall-back transition is biologically easier, since most people drift later naturally. The American Academy of Sleep Medicine has published a position statement in favor of permanent standard time over the current biannual DST shift.
Weekend social jet lag. Coined by the Roenneberg group in the mid-2000s, social jet lag describes the misalignment between the body's chronotype and the schedule imposed by work or school days. Caliandro and colleagues' 2021 review reports that approximately 70% of students and workers experience at least one hour of social jet lag, and nearly half experience two hours or more. Social jet lag is associated with elevated rates of obesity, type 2 diabetes, smoking, and depression. The catch-up question — whether sleeping in on weekends repairs the damage — is covered in Can You Catch Up on Sleep on Weekends?.
Late-bedtime drift. Adolescents and young adults have a chronotype that runs naturally later, and exam periods, gaming, and social media accelerate the drift. The reset to an earlier schedule at the start of a school term or new job is the most common civilian sleep-schedule reset.
Shift work. AASM materials suggest that approximately one in five shift workers meets the diagnostic criteria for shift work sleep disorder. The published interventions and chronobiology of shift work are a clinical topic outside the scope of an informational guide; consultation with occupational medicine or sleep medicine is the standard professional path.
Evidence-based intervention strategies
1. Gradual shift (~30–60 minutes per day) is more reliable than aggressive shifts. Eastman and colleagues' 2005 study in SLEEP compared 1-hour-per-day and 2-hour-per-day pre-flight phase advances with intermittent morning bright light over three days. Their conclusion: "sleep should be advanced by 1 hour per day but not 2 hours per day, as advancing 2 hours per day may make falling asleep at night difficult." The 1-hour-per-day protocol produced a stable phase advance of approximately 1.4 hours over three days.
2. Light position is decisive. Light at the wrong circadian phase produces effects in the wrong direction. The PRC-aligned defaults:
- For phase advance — direct sunlight or a ≥10,000-lux box for 30 minutes to 2 hours immediately on waking; avoid bright light in the evening.
- For phase delay — bright light in the evening (or a light box if natural light is unavailable); avoid bright morning light.
3. Caffeine — both the use and the cutoff matter. CDC guidance describes 200 mg of caffeine every four hours, stopped at least six hours before bedtime, as a useful daytime alertness adjunct. Burke and colleagues' 2015 paper in Science Translational Medicine showed that caffeine equivalent to a double espresso consumed three hours before bedtime delayed the circadian melatonin rhythm by approximately 40 minutes — about half the magnitude of a 3,000-lux evening light pulse. Caffeine is itself a chronobiotic, in other words, and late afternoon use can drift the clock in the wrong direction during a phase-advance reset. The companion guide on this site, How Long Before Bed Should You Stop Drinking Caffeine?, covers cutoff timing in depth.
4. Sleep restriction as an adjunct. Temporarily compressing the sleep window raises homeostatic sleep pressure and helps consolidate sleep at the new target time. This is a CBT-i component, distinct from a single all-nighter — sleep restriction is a planned, sustained reduction of time in bed, with the goal of strengthening sleep drive at the desired phase. A standalone all-nighter does not move the SCN clock; sleep restriction over multiple nights, paired with correctly timed light, can support a reset.
5. Pre-shift adaptation for travel. For an eastbound flight, advance bedtime and wake time by 30–60 minutes per day for the three days before departure, with bright light immediately on waking. Burgess, Crowley, Gazda, Fogg, and Eastman's 2003 protocol in SLEEP is the standard published reference for the pre-flight intervention.
Four 7-day plan templates (informational, not prescriptive)
The following templates are informational examples of how the principles above translate into daily steps. They are not medical recommendations. Anyone with underlying conditions, on medications affecting sleep, or pregnant should consult a clinician before deliberately shifting their sleep schedule.
Plan A — Phase advance (need to wake earlier; e.g., new school term, job change).
| Day | Bedtime shift | Wake shift | Light | Notes |
|---|---|---|---|---|
| 1 | −30 min | −30 min | Direct sunlight 15–30 min within 30 min of waking | Avoid bright screens after sunset |
| 2 | −60 min | −60 min | Same; outdoor walk preferred | |
| 3 | −90 min | −90 min | Same; consider 10,000-lux box if no sun | Caffeine cutoff = bedtime − 6 hours |
| 4 | −2 h | −2 h | Same | |
| 5 | −2.5 h | −2.5 h | Same | |
| 6 | −3 h (target) | −3 h (target) | Same | |
| 7 | Hold target | Hold target | Same | Consistency matters more than perfection |
Plan B — Phase delay (need to stay up later). The mirror image of Plan A. Shift bedtime and wake time +30 to +60 minutes per day later, avoid morning light (sunglasses outdoors, dim room), seek bright light in the evening. Less commonly needed as a personal goal, but the same principles apply.
Plan C — Eastward jet lag (e.g., LAX → London, ~8 hours east). Pre-flight: starting three days before departure, advance bedtime and wake time by 1 hour per day, with bright light immediately on waking. On arrival: morning sunlight at the destination, avoid evening light, consider low-dose melatonin (~0.3–0.5 mg) about 5 hours before the local target bedtime for the first 2–3 nights. Expect roughly 1 hour per day of natural circadian adaptation; full re-entrainment may take a week or more.
Plan D — Westward jet lag (e.g., London → LAX, ~8 hours west). Easier, because the body drifts later naturally. On arrival: stay awake at the destination until the local bedtime, seek evening sunlight, avoid intense morning light for the first 1–2 days. Expect roughly 1.5 hours per day of natural adaptation; most people fully adjust within 3–5 days.
Once a target bedtime is fixed, the Sleep Cycle Calculator can map it onto cycle-aligned options so the alarm catches the end of a 90-minute cycle rather than mid-cycle. The pairing of a planned reset (this article) with cycle-aligned wake (How Sleep Cycles Work) is the kind of integration the cluster is built for.
What does not work or is over-marketed
The single all-nighter "reset" is a myth. Twenty-four to thirty-six hours of wakefulness raises homeostatic sleep pressure (adenosine accumulation), so the next sleep onset is faster. The SCN clock, however, does not move under sleep deprivation alone — it remains where the prior light/melatonin signals placed it. Without aligned zeitgebers in the days that follow, the body drifts back to its prior schedule. Sleep deprivation can also disturb clock-gene expression in peripheral tissues for days afterward, an extra metabolic cost without a corresponding clock benefit.
Untimed light boxes do little useful. A 10,000-lux box used "whenever it's convenient" can shift the clock the wrong way if used in the evening. The phase response curve is the precondition for benefit; a light box without timing is equivalent to medication without dosing.
High-dose melatonin (5–10 mg) produces little additional phase shift versus 0.3–0.5 mg, with worse next-day side-effect profiles. The Erland and Saxena 2017 product-variability finding (label values from −83% to +478% of measured) means a "5 mg" pill could deliver 1 mg or 27 mg in practice — even setting aside whether 5 mg is the right intended dose.
"Just go to bed earlier" without zeitgeber alignment. Lying in bed during the body's evening alertness peak (the so-called "forbidden zone," 2–3 hours before DLMO) tends to produce frustrating wakefulness, which over time conditions the bed as a place of arousal rather than sleep. The companion guide How to Fall Asleep Faster covers stimulus control and the AASM behavioral treatment guideline in detail.
Blue-light blocking glasses. A 2025 Frontiers in Neurology meta-analysis pooled actigraphy-based randomized trials of blue-blocking glasses and reported a sleep-onset-latency mean difference of −4.86 minutes, not statistically significant. A 2024 Translational Vision Science & Technology analysis found that many commercially marketed "blue-blocking" glasses lack the melanopic daylight filtering density (mDFD ≥1) that would justify the label. The mechanism is plausible; the measured effect on sleep is small or null.
The Argonne anti-jet-lag diet. Developed by Charles Ehret at Argonne National Laboratory in the 1980s — a feast/fast/feast/fast cycle starting four days before travel, with structured caffeine timing. Reynolds and Montgomery's 2002 study in Military Medicine reported lower jet lag rates among diet users in a deployed military population (N=186, self-selected observational design, not a randomized controlled trial). The protocol has not been adopted into modern chronobiology guidelines; light and melatonin timing are the dominant evidence-based interventions. The Argonne diet is mostly of historical interest at this point.
When clinical evaluation is described as warranted
This section is informational, not diagnostic. Patterns the clinical literature describes as commonly warranting evaluation by a sleep-medicine clinician include:
- Two or more weeks of sustained effort on the principles above with no measurable shift toward the target schedule.
- Suspected DSPD/DSWPD (delayed sleep–wake phase disorder) — chronic inability to fall asleep before approximately 2–6 AM, with significant functional impairment when forced to wake earlier (most common in adolescents and young adults).
- Suspected ASPD/ASWPD (advanced sleep–wake phase disorder) — overwhelming evening sleepiness around 6–9 PM, with awakening between 2 and 5 AM (more common in older adults).
- Shift work sleep disorder — microsleeps during night shifts, daytime sleep under 4 hours, accident risk awareness.
- Suspected non-24 sleep-wake disorder — particularly in totally blind individuals, where studies report up to 70% prevalence — characterized by daily phase drift.
- Concurrent conditions — depression, bipolar disorder, ADHD, parasomnias, suspected sleep apnea — sleep-medicine evaluation is the standard professional pathway when these overlap with persistent schedule misalignment.
Diagnostic tools used by clinicians include actigraphy, two-week sleep diaries, and DLMO measurement (typically in research or specialty centers). Treatment is usually a combination of light therapy, low-dose melatonin, and chronotherapy (planned schedule shift) per the AASM 2015 clinical practice guideline for circadian rhythm sleep–wake disorders.
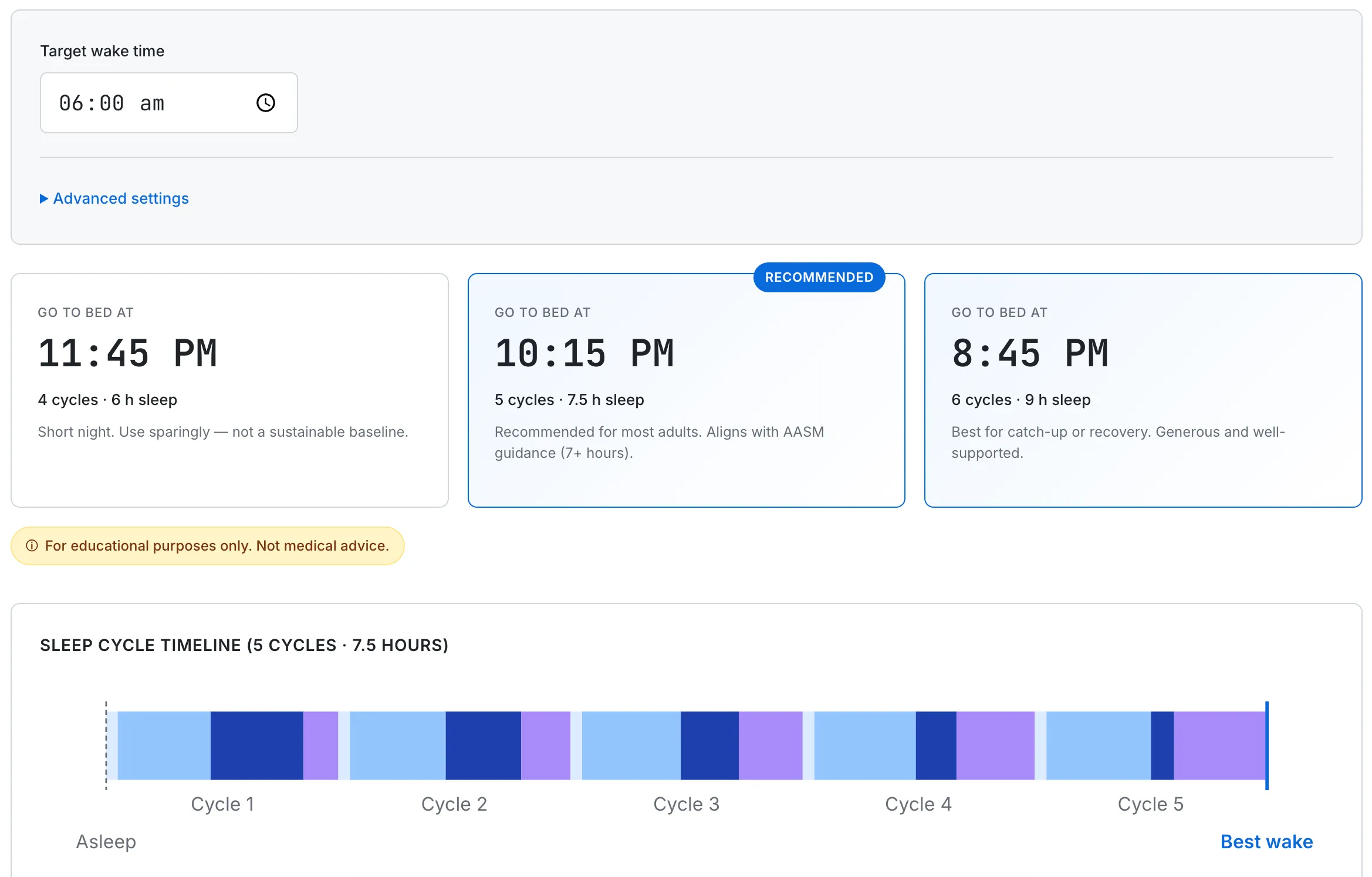
Once a target bedtime is set, mapping it onto a cycle-aligned wake time helps the alarm catch the end of a 90-minute cycle rather than mid-cycle — useful when locking in the new schedule after a planned reset.
Common questions about resetting a sleep schedule
Can I reset my sleep schedule in one night?
The published evidence does not support a single-night reset. Twenty-four hours of wakefulness raises homeostatic sleep pressure (adenosine accumulation), so the next sleep onset comes faster — but the SCN clock does not move under sleep deprivation alone. Without aligned light and melatonin signals, the body drifts back to its prior schedule within days. The studied interventions involve gradual phase shifts of 30–60 minutes per day combined with morning bright light (for phase advance) or evening light (for phase delay).
How long does it take to reset your sleep schedule?
The CDC's Yellow Book 2026 chapter on jet lag describes natural adaptation rates of roughly 1 hour per day eastward and 1.5 hours per day westward. For a 1-hour shift in either direction, expect about a day; for a 3-hour shift, roughly 2–3 days westward or 3 days eastward. Pre-flight or pre-shift interventions can compress this somewhat by starting the shift before the new schedule begins.
Is the all-nighter reset method effective?
No. Sleep deprivation moves homeostatic pressure but not the circadian clock. Without aligned light and melatonin in the days that follow, the body drifts back. Sleep deprivation can also disturb peripheral clock-gene expression for days afterward, adding metabolic cost without a corresponding clock benefit.
Does melatonin help reset your sleep schedule?
Low-dose melatonin (≈0.3–0.5 mg) timed correctly can shift the circadian clock by up to about 1.5 hours per dose (Burgess, Revell, Eastman 2010). For phase advance, melatonin is taken in the late afternoon or early evening, roughly 5–7 hours before the desired bedtime. Higher doses (5–10 mg) do not produce larger phase shifts and tend to produce more next-day side effects. Erland and Saxena 2017 documented that label and measured content of commercial melatonin products diverge by −83% to +478%, complicating dose precision. Regulatory status varies internationally.
What is the best way to fix a delayed sleep phase?
The published combination is: bright light immediately on waking (direct sunlight or a 10,000-lux box for 30 minutes to 2 hours), avoidance of bright light in the evening, optionally low-dose melatonin in the late afternoon or early evening (5–7 hours before target bedtime). Gradual phase advance of 1 hour per day, sustained over 5–7 days, produces a more stable shift than a 2-hour-per-day jump.
How do you fix jet lag?
Direction matters. Eastward jet lag requires phase advance and is biologically harder; westward jet lag requires phase delay and is easier. The CDC's published guidance describes 0.5–1 mg of melatonin as often sufficient for circadian shifting; 200 mg of caffeine every 4 hours during the day with a 6-hour pre-bedtime cutoff as a useful daytime alertness adjunct; and natural adaptation rates of roughly 1 hour per day eastward, 1.5 hours per day westward. Pre-flight phase shifting (advancing or delaying bedtime by 1 hour per day for 3 days before departure) compresses post-flight adaptation time.
Do blue-light glasses help reset your sleep schedule?
The published evidence is weak. A 2025 Frontiers in Neurology meta-analysis of randomized trials reported a pooled sleep-onset-latency mean difference of −4.86 minutes, not statistically significant. A 2024 TVST analysis found that many marketed "blue-blocking" glasses lack the melanopic daylight filtering density that would justify the label. They are at best a small adjunct; the dominant interventions are light timing and low-dose melatonin.
This guide summarizes circadian science and the published evidence on phase-shifting interventions, drawn from primary research (Czeisler 1999, Khalsa 2003, Burgess & Eastman 2010, Eastman 2005, Burke 2015), authoritative guidelines (AASM 2015, CDC Yellow Book 2026), and recent systematic reviews. Circadian science evolves and individual responses vary widely with age, chronotype, medications, and underlying conditions. None of this is medical advice or a substitute for professional consultation. For persistent schedule misalignment, suspected sleep disorders, or concurrent mental-health conditions, please consult a licensed sleep-medicine professional.