Are Naps Good or Bad for Adults? What Research Shows
Brief afternoon naps look helpful in controlled studies. Long habitual naps track with cardiovascular risk. The honest read on what the literature actually shows — and what it doesn't.
This is an informational summary of peer-reviewed sleep research and clinical literature on napping. It is not medical advice, diagnosis, or treatment guidance. The studies cited describe controlled lab conditions or population cohorts — not personal predictions, and individual nap response varies with age, sleep history, medications, and underlying conditions. If persistent daytime sleepiness, loud snoring, or unrefreshing sleep is affecting your day, please consult a licensed healthcare professional.
In 1995, NASA published a study that aviation medicine has cited often since. Twelve long-haul pilots were given a 40-minute nap opportunity during the cruise phase; nine other pilots were instructed to stay awake as usual. The nap group fell asleep on 93% of opportunities, took an average of 5.6 minutes to drift off, and slept for an average of 25.8 minutes. The decisive comparison happened during the most demanding phases — descent and landing — where the nap group was measurably more alert and had fewer attention lapses. The result reshaped how aviation handles fatigue, and it reframed a question many adults quietly ask themselves — is napping a useful tool, or a habit that's quietly working against me?
The short answer. Brief naps — roughly 10 to 20 minutes — are consistently associated in controlled studies with improvements in alertness, reaction time, and short-term memory, especially in sleep-deprived participants (Brooks & Lack 2006; the NASA Rosekind 1995 work). Past about 30 minutes, the risk of sleep inertia — that 30-to-60-minute foggy window after waking from deep sleep — climbs sharply. Habitual naps longer than an hour are observationally linked to higher cardiovascular and all-cause mortality — although that long-nap signal appears to be partly confounded by underlying illness rather than caused by long napping itself (Yamada 2015; Liu 2024). Naps after about 3 PM, or longer than 60 minutes, tend to delay nighttime sleep onset for most adults. The honest summary — for a well-rested adult, a brief early-afternoon nap is low-risk and often helpful; habitual long naps without an obvious sleep deficit are a signal worth paying attention to.
This post walks through (1) what brief naps actually do for the brain and why pilots nap, (2) the 10/30/90-minute nap trade-off, (3) the post-lunch dip and the natural nap window, (4) the honest read on habitual long naps and the cardiovascular research, (5) the coffee nap — what Reyner & Horne and Hayashi actually showed, and (6) when daytime sleepiness is described in the literature as a clinical signal.
What naps actually do for the brain (and why pilots nap)
The Rosekind 1995 work serves as an anchor because it produced controlled data under real long-haul flight conditions. The nap group (n=12) fell asleep on 93% of opportunities, with an average sleep latency of 5.6 minutes and an average sleep duration of 25.8 minutes. NASA-affiliated reports of the study cite alertness and psychomotor vigilance gains of roughly 54% and 34% respectively versus the no-nap control, with effects sustained through the descent and landing phases. The 25.8-minute average was a measured outcome, not a recommendation — and the reason later research describes shorter nap lengths as more reliably useful becomes clearer in the next study.
Brooks & Lack 2006 directly compared 5-, 10-, 20-, and 30-minute afternoon naps against a no-nap control after a night of sleep restriction. The 10-minute nap produced immediate improvements across every outcome — subjective sleepiness, cognitive performance, and alertness — with effects sustained for roughly 155 minutes. The 30-minute nap showed sleep inertia immediately after waking, and improvements only appeared in the same window after that inertia faded. Five minutes was too short to register much. The literature on brief naps after partial sleep loss converges on this pattern: shorter is more reliable than the 25-to-30-minute range.
At the longer end, the Mednick research line tells a different story. Mednick 2002 (PMID 12032542, Nature Neuroscience) compared 30- and 60-minute midday naps and showed that 60-minute naps rich in slow-wave sleep reversed the perceptual deterioration that builds up across a day of testing. The follow-up Mednick 2003 (PMID 12819785, Nature Neuroscience) reported that 60-to-90-minute naps containing both slow-wave sleep and REM matched 8-hour overnight sleep on the same perceptual task — the basis for the "a nap is as good as a night" framing. A subsequent PNAS paper (Cai, Mednick et al. 2009) reported that nap groups containing REM sleep improved by roughly 40% on the Remote Associates Test relative to morning baseline, evidence the literature describes as REM-specific creative consolidation. The UPenn group led by Dinges has shown that a 2-hour prophylactic nap reduces microsleeps and performance decrements during sustained wakefulness even 12 hours later.
The mechanism that makes the 90-minute "full-cycle nap" possible is covered in depth in How Sleep Cycles Work.
The 10/30/90-minute trade-off — what your nap length wakes you from
The trade-off in nap length is, in practical terms, a question about which stage you're most likely to wake up from. Waking from N1 or early N2 and waking from the middle of N3 are, for the same person, very different experiences.
| Length | Sleep stages reached | Cognitive effect | Sleep inertia risk | Best for |
|---|---|---|---|---|
| 10–20 min | Mostly N1 and early N2 | Improved alertness, reaction time, vigilance within minutes; benefits sustained 1–2.5 hours (Brooks & Lack 2006) | Low — waking from light sleep | Workday refresh; pre-meeting alertness; drivers (with caffeine — Reyner & Horne 1997) |
| 30–60 min | Includes N3 (deep slow-wave sleep) | Declarative memory consolidation; longer-lasting alertness gains; but a measurable 30-to-60-minute post-nap performance dip (sleep inertia) | High — waking from N3 produces a measurable performance drop (Hilditch & McHill 2019, citing Stampi) | Recovery from sleep restriction, when the post-nap window allows buffer time |
| ~90 min | Full cycle: N1→N2→N3→REM, ending at a light stage | REM-linked associative and creative consolidation (Mednick 2003; Cai 2009); perceptual learning equivalent to a full night for some tasks | Low — wakes near the end of a cycle | Catch-up after a clearly short night; weekend recovery; emergency and shift workers (Sleep Foundation) |
The reason the 30-minute zone is the trickiest sits in stage architecture. Stage N2 typically begins at around 25 minutes in the first cycle and lengthens across the night, accounting for roughly 45% of total sleep; N3 (slow-wave sleep) is longer in early cycles and shortens as the night progresses (StatPearls). A 30-to-60-minute nap is precisely the window in which a sleeper is most likely to be woken from the middle of N3.
A 2019 review (Hilditch & McHill) cites Stampi's earlier work showing roughly a 41% performance reduction immediately after waking from slow-wave sleep, with cerebral blood flow remaining below pre-sleep levels for about 30 minutes. That same sleep-inertia mechanism is also why an alarm that happens to ring mid-deep-sleep can leave someone feeling worse after eight hours than after a clean 7.5 — covered in detail in Why Am I Tired After 8 Hours of Sleep?
The post-lunch dip — why early afternoon is the natural nap window
The drowsiness many adults notice between roughly 1 and 3 PM isn't only about lunch. Monk 2005 explicitly reviewed evidence that a "post-lunch dip in performance" is observed even when participants skip lunch — meaning the drop is circadian, not purely a meal effect.
Three mechanisms overlap. First, homeostatic adenosine pressure that accumulates while awake — increasing gradually over hours since wake. Second, an early-afternoon trough in the circadian "wake-promoting signal" — a pattern consistent with biomathematical sleep-propensity modeling (Bes et al. 2009, Sleep). Third, a small dip in core body temperature. Adenosine pharmacology is well mapped in lab work — adenosine accumulates during wake and binds A1 and A2A receptors to drive sleep pressure; caffeine antagonizes the same receptors (Ribeiro & Sebastião 2010).
The "avoid napping after about 3 PM" rule of thumb follows from that mechanism. The Sleep Foundation describes a safe nap window as ending roughly 8 hours before bedtime — for an 11 PM bedtime, before about 3 PM. Mayo Clinic similarly describes the early-afternoon window, around 2 to 3 PM, as preferable. Cousins et al. 2022 reported that in healthy young adults, late-afternoon and evening naps delayed nighttime sleep onset and reduced quality markers of the subsequent night's sleep.
Are habitual long naps bad for you? The honest read on the cardiovascular research
Every association in this section comes with a reverse-causation caveat. The data are real, but a single number pulled out of context produces a misleading causal frame.
The Yamada 2015 meta-analysis (SLEEP, 11 prospective cohorts, n=151,588, mean follow-up 11 years) reported a J-shaped curve. Naps under 60 minutes per day showed essentially no association — CVD relative risk 1.00 (95% CI 0.84–1.18); all-cause mortality RR 1.07 (0.99–1.16). Risk dipped slightly in the 0-to-30-minute range, ticked up around 45 minutes, and rose sharply past 60 minutes. At 60+ minutes: CVD RR 1.82 (1.22–2.71); all-cause mortality RR 1.27 (1.11–1.45). The authors flagged reverse causation in the paper itself — long nappers "could be more likely to be ill and have various risk factors."
The Liu 2024 meta-analysis (PLOS ONE, 21 cohort studies, n=371,306, follow-up 4–18 years, 11 countries) reproduced the same pattern in a larger sample. Nappers vs non-nappers: all-cause mortality HR 1.28 (1.18–1.38). Under 1 hour: HR 1.00 — not statistically significant. One hour or more: all-cause mortality HR 1.22 (1.12–1.33); CVD HR 1.37 (1.09–1.71). The authors directly raised the confounding hypothesis — "physically weak individuals have significantly reduced daytime activity and increased time spent in bed." That is, the data don't rule out a scenario in which long naps don't cause cardiovascular disease but in which already-developing-but-undiagnosed disease produces fatigue, and that fatigue drives the long naps. Underlying mechanisms — sleep apnea, depression, chronic illness — that independently raise daytime sleep pressure are covered in Why Am I Tired After 8 Hours of Sleep?
A frequently cited finding pointing the other way comes from Naska 2007 (the Greek arm of EPIC, Archives of Internal Medicine, 23,681 Greek adults, ~6 years of follow-up). The paper reported a 12% lower coronary mortality rate among occasional siesta-takers and a 37% lower rate among systematic siesta-takers (with a larger 64% reduction in working men). Subsequent analyses described a more mixed picture — a Mediterranean lifestyle context, lower occupational stress, and other protective factors are difficult to disentangle from the nap itself. This study is cited here not as a lifestyle endorsement but to show that confounding works in both directions.
A Mendelian randomization analysis on UK Biobank data (Dashti et al. 2021, Nature Communications, n=452,633, 123 nap-associated genetic loci) suggested a potentially causal link between more frequent napping and higher blood pressure and waist circumference — although the estimated causal effect sizes were small. The AASM editorial "Naps and cardiovascular disease: you snooze, you lose!" reviews the broader literature in the reverse-causation context.
The closing message of this section — short naps were not linked to cardiovascular outcomes in the same studies. The point at which the research describes a real signal is habitual naps of an hour or more, and even that signal is partly explained by reverse causation. None of these statistical patterns are personal predictions.
The coffee nap — what Reyner & Horne and Hayashi actually showed
The coffee-and-nap combination — the "coffee nap" — is often overstated online, but the lab-data footprint is small and consistent.
Pharmacology first. Caffeine has an average plasma half-life of approximately 5 hours (range 1.5–9.5 hours), and peak plasma concentrations are reached between 15 and 120 minutes after ingestion (IoM, Pharmacology of Caffeine). Approximately 75–80% is metabolized to paraxanthine, which itself binds adenosine receptors with similar affinity. Caffeine is a non-selective A1, A2A, A2B, and A3 antagonist; A2A receptor blockade is commonly cited as the principal wake-promoting mechanism (Ribeiro & Sebastião 2010).
Reyner & Horne 1997 (Psychophysiology) randomized 12 sleepy participants across three conditions during a 30-minute break — (a) 200 mg caffeine plus a short nap, (b) caffeine alone, or (c) placebo. Participants then drove a monotonous afternoon driving simulator for two hours. The frequency of driving incidents relative to placebo was: placebo 100%, caffeine alone 34%, and caffeine plus a short nap roughly 9%. Subjective sleepiness and EEG-based sleepiness markers tracked the same pattern.
Hayashi 2003 (Clinical Neurophysiology) tested 10 young adults across five conditions — no nap; nap only; caffeine plus nap; nap with bright light; nap with face washing. Caffeine taken immediately before a 20-minute nap was the most effective combination for both subjective sleepiness and task performance, with effects sustained for about an hour after the nap.
The mechanism is two compounding effects. Caffeine takes about 20 to 30 minutes to cross the blood-brain barrier and antagonize adenosine receptors. A 15-to-20-minute nap immediately after coffee uses that interval to lower natural sleep pressure with a brief sleep, and the caffeine's wake effect comes online right at the moment of waking.
An honest caveat. Most coffee-nap evidence comes from small samples (n < 20), short-term, and in participants who were already sleepy or specifically sleep-deprived. For real fatigue scenarios — long-distance driving, post-shift recovery — the lab evidence is robust. As a routine lifestyle habit for offsetting deliberate sleep restriction, it is not validated.
When daytime sleepiness is described as a clinical signal in the literature
This section doesn't provide diagnostic guidance. It summarizes patterns the clinical literature describes as signals, and the general points at which escalation is described as appropriate.
Obstructive sleep apnea (OSA). Daytime sleepiness driving naps is a documented hallmark of untreated OSA. The STOP-BANG questionnaire is a widely used validated OSA screening tool (Chung et al.). When loud snoring, witnessed apneas, and unrefreshing nighttime sleep co-occur, the literature commonly describes a clinical evaluation as warranted.
Depression. Depression is associated bidirectionally with sleep disturbance — some patterns include hypersomnia, while others present with insomnia and early-morning awakening (Riemann et al., review). When sustained low mood pairs with two-or-more weeks of changed daytime sleep, the literature commonly describes that combination as worth a clinical evaluation.
Narcolepsy. The cardinal symptom is excessive daytime sleepiness. Supporting features include sudden sleep attacks, cataplexy (muscle-tone loss with strong emotion), sleep paralysis, and hypnagogic hallucinations (NHLBI). Clinical reviews of narcolepsy describe brief naps as characteristically more refreshing in narcolepsy than in other sleepiness disorders. A diagnostic note — OSA can mimic features of narcolepsy in some cases, including producing false-positive findings on diagnostic testing (case report, PMID 30523556).
Long or frequent napping in older adults. The 2022 UCSF communication "Extended napping in seniors may signal dementia" summarized a study reporting that excessive daytime sleep in older populations may be an early signal of cognitive decline. Clinical reviews of older-adult napping and broader systematic reviews of sleep and cognitive health share similar framing.
Key framing for older adults — long or frequent napping in older people often reflects fragmented nighttime sleep (sleep apnea, restless legs, nocturia, and similar). The consensus in the literature is closer to "investigate the night" than to "sleep needs drop with age." A pattern where early-morning waking drives daytime napping has its own diagnostic shape, covered in Why Do I Wake Up at 3 AM Every Night?
For sleep-deficit recovery contexts where naps are weighed against weekend catch-up sleep — including the limits of "marathon naps" and weekend make-up — the framing in Can You Catch Up on Sleep on Weekends? covers the trade-offs.
What's the perfect nap length?
For most adults, controlled studies describe roughly 10 to 20 minutes as the sweet spot — long enough to reduce sleepiness and improve reaction time, short enough to avoid the sleep inertia that comes from waking out of deep sleep (Brooks & Lack 2006). The next clean option is around 90 minutes — completing a full cycle and waking near a lighter stage similar to the start. The 30-to-60-minute zone is, for many adults, the foggiest length to wake up from.
Is a 30-minute nap good?
The literature describes it as mixed. A 30-minute nap can produce a longer alertness boost than a 10-minute nap, but it's also a length where many adults are starting to enter deep slow-wave sleep — meaning the 30-to-60-minute period after waking can feel foggier than before the nap (sleep inertia). If 30 minutes of buffer time before a focus-demanding task is guaranteed, the trade-off is acceptable. If not, 10 to 20 minutes is described as the lower-risk option.
Are naps after 3 PM bad?
For most adults trying to sleep at a normal nighttime hour, the literature describes them that way. The Sleep Foundation describes a safe nap window as ending about 8 hours before bedtime — for an 11 PM bedtime, before roughly 3 PM. Late-afternoon and evening naps reduce the adenosine pressure that drives sleep onset, and controlled studies have linked them to delayed nighttime sleep onset and reduced sleep-quality markers (Cousins 2022).
Why do I feel worse after a nap?
Almost always sleep inertia — waking from the middle of a deep N3 stage. The brain doesn't wake uniformly; the brainstem wakes first and the prefrontal cortex last, and cerebral blood flow remains below pre-sleep levels for about 30 minutes (Hilditch & McHill 2019). The literature describes two ways to avoid that window — a brief nap (10–20 minutes) that ends before deep sleep starts, or a roughly 90-minute nap that finishes a full cycle and wakes during a lighter stage.
Are long naps bad for the heart?
Observationally, habitual naps of 60 minutes or more are associated with higher cardiovascular and all-cause mortality risk (Yamada 2015 meta-analysis: ≥60 min vs no nap, CVD RR 1.82; Liu 2024: ≥1 hr CVD HR 1.37). That association is at least partly confounded — people with chronic illness tend to nap longer because of disease-driven fatigue, rather than the napping causing the disease. Naps under an hour were not linked to cardiovascular risk in the same studies. None of these statistical patterns are personal predictions.
Do coffee naps actually work?
For acute sleepiness, small controlled studies consistently report yes. In Reyner & Horne 1997's driving simulator study, 200 mg caffeine plus a short nap reduced lane-departure events to roughly 9% of the placebo level — a larger effect than caffeine alone (34%) or no caffeine. Caffeine takes 20 to 30 minutes to reach adenosine receptors, which aligns with a brief nap length. The lab evidence in real sleep-pressure scenarios — long drives, post-shift recovery — is robust; routine use as an everyday lifestyle habit is not validated.
Should older adults nap?
Brief, intentional afternoon naps (under 30 minutes) are described in the literature as generally fine, and may be supportive when nighttime sleep is fragmented. Long, frequent, or unintentional daytime sleep in older adults, however, is more often described as an underlying signal — sleep apnea, restless legs, depression, or in some research an early signal of cognitive decline (UCSF 2022). The general framing in the literature points toward investigating nighttime sleep rather than expanding daytime napping.
If a 90-minute full-cycle nap is on the plan, the Sleep Cycle Calculator shows the exact end time within the early-afternoon window where the cycle finishes cleanly.
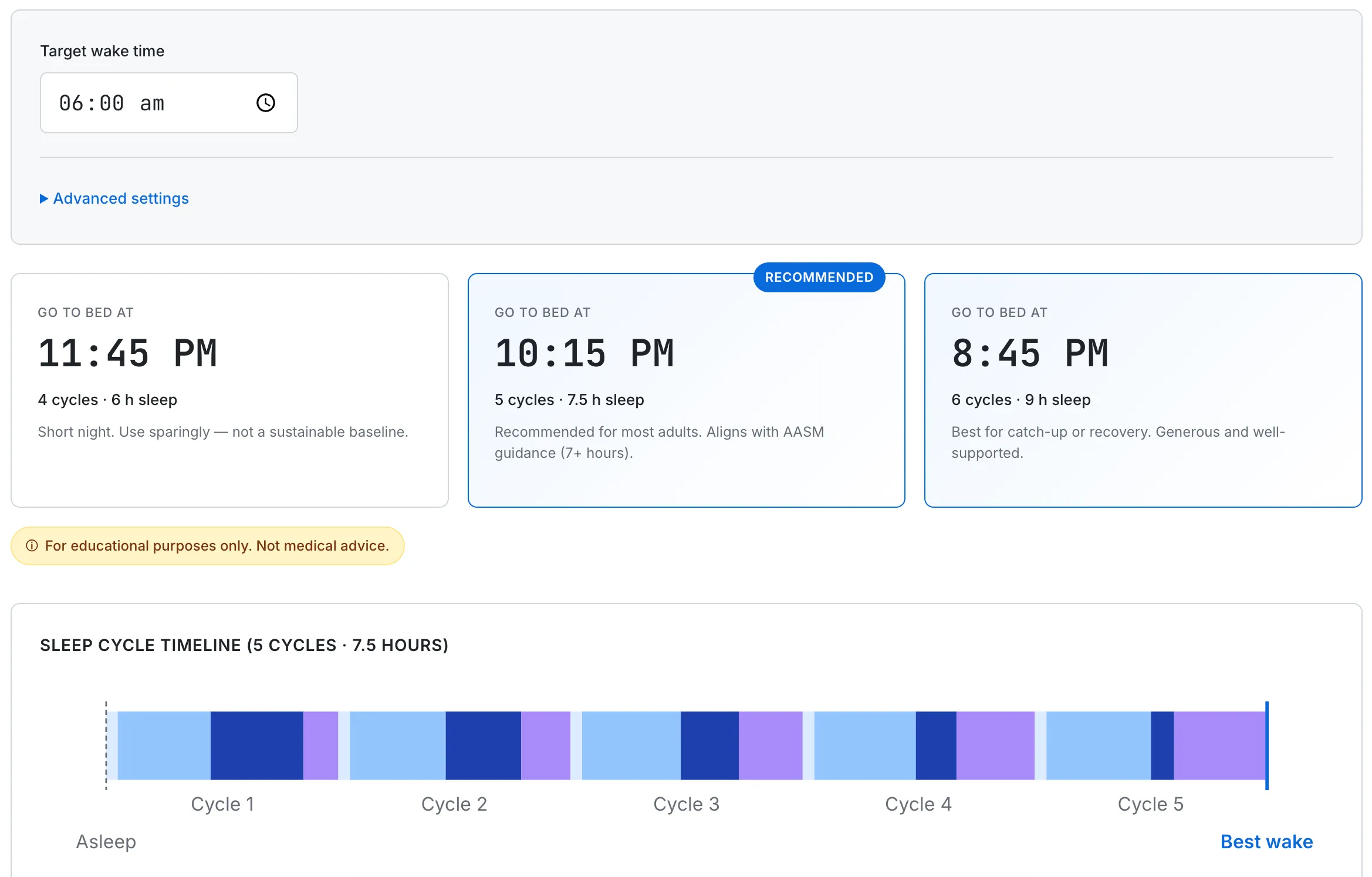
A 90-minute full-cycle nap walks through N1→N2→N3→REM and ends near a lighter stage, similar to where it started. Cycle length and onset time are tunable for individual variation.
This guide summarizes general adult sleep medicine drawn from peer-reviewed sources, and is for educational use only. The long-nap and cardiovascular associations described above are observational and partly confounded by underlying illness — they describe statistical patterns at the population level, not personal predictions. None of this is medical advice or a substitute for a clinical evaluation. If you have ongoing sleep difficulty, suspect a sleep disorder, or are concerned about excessive daytime sleepiness, please consult a licensed healthcare professional.